16.10.2014
For the first time, researchers have succeeded to detect a single hydrogen atom using magnetic resonance imaging, which signifies a huge increase in the technology's spatial resolution. In the future, single-atom MRI could be used to shed new light on protein structures.
Conventional magnetic resonance imaging (MRI), well-known from its use in hospitals, can typically resolve details of up to one tenth of a millimetre, for example in cross-sectional images of the human body. Together with colleagues at the University of Leipzig, researchers of ETH Zurich are working on massively increasing the resolution of the technique, with the goal of eventually imaging at the level of single molecules – demanding an over one million times finer resolution. By detecting the signal from a single hydrogen atom, they have now reached an important milestone toward such single-atom MRI.
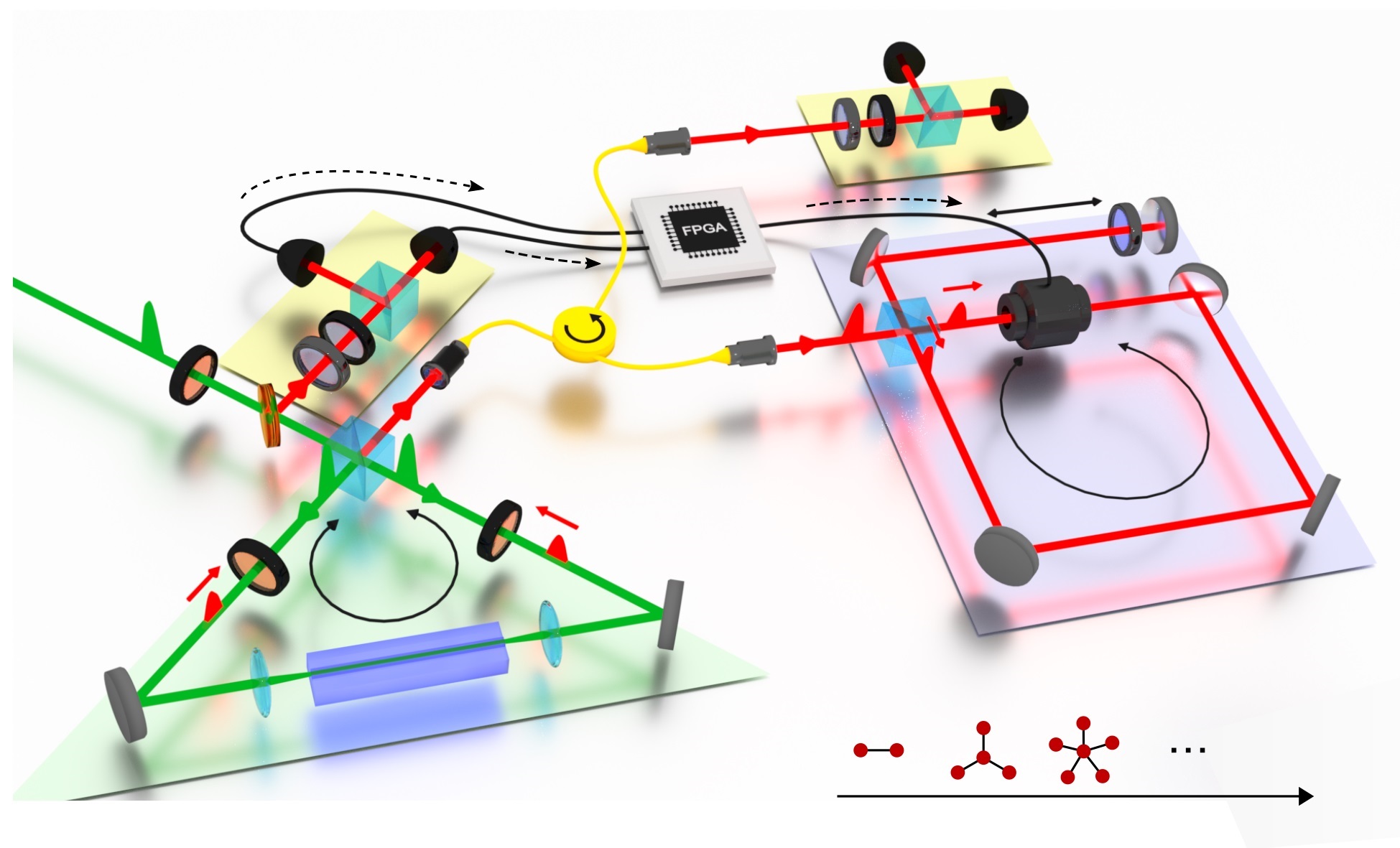
The research team led by Christian Degen, Professor at the Laboratory for Solid State Physics, developed a different and vastly more sensitive measurement technique for MRI signals. In standard hospital instruments, the magnetisation of the atomic nuclei in the human body is inductively measured using an electromagnetic coil. "MRI is nowadays a mature technology and its spatial resolution has remained largely the same over the last ten years. Physical constraints preclude increasing the resolution much further," explains Degen. In their experiments, the ETH researchers measured the MRI signal with a novel diamond sensor chip using an optical readout in a fluorescence microscope.
Diamond impurity formed the sensor
The sensor consisted of an impurity in diamond known as the nitrogen-vacancy centre. In this case, two carbon atoms are missing in the otherwise regular diamond lattice, while one of them is replaced by a nitrogen atom. The nitrogen-vacancy centre is both fluorescent and magnetic, making it suitable for extremely precise magnetic field measurements.
For their experiment, the researchers prepared an approximately 2x2 millimetre piece of diamond such that nitrogen-vacancy centres formed only a few nanometres below the surface. By an optical measurement of the magnetisation, they were in several cases able to confirm the presence of other magnetic atomic nuclei in the immediate vicinity. "Quantum mechanics then provides an elegant proof of whether one has detected an individual nucleus, or rather a cluster of several hydrogen atoms," states Degen. The researchers also used the measured data to localize the hydrogen nuclei with respect to the nitrogen-vacancy centre with an accuracy of better than one angstrom (one ten-millionth of a millimetre).
Nano-MRI for structural biology
"This is an important intermediate step toward the mapping of entire molecules," explains the ETH professor, who was awarded an ERC starting grant by the European Research Council in 2012 for his research in this field. In a next step, the researchers envision imaging a small molecule with their nano-MRI device. Even if it would become possible to map a large number of atoms, however, it is neither the aim nor practical to investigate an entire human body at atomic level with this technology.
The dream of the researchers is to one day apply the technology to shed light on the spatial structure of biomolecules, such as proteins. At present, researchers usually rely on X-ray crystallography to investigate protein structures. This requires growing crystals consisting of billions of identical molecules. Crystallising proteins is, however, often very challenging, and sometimes impossible. If the ETH physicists achieve their goal, a single molecule would in principle suffice for determining the structure. Another advantage of nano-MRI is that the molecules can be labelled by isotopes, providing a means for site-specific image contrast. This would help biologists tackle issues relating to protein functions more effectively.
Literature reference
Loretz M, Rosskopf T, Boss JM, Pezzagna S, Meijer J, Degen CL: Single proton spin detection by diamond magnetometry. Science, online publication 16 October 2014, doi: 10.1126/science.1259464